Featured Products

Maximise your skillset with minimally invasive care

money4dentists wins another award!

A new lease of life

Take your practice to another level

Exploring implantology with the ADI
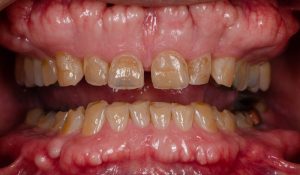
Tackling tooth wear
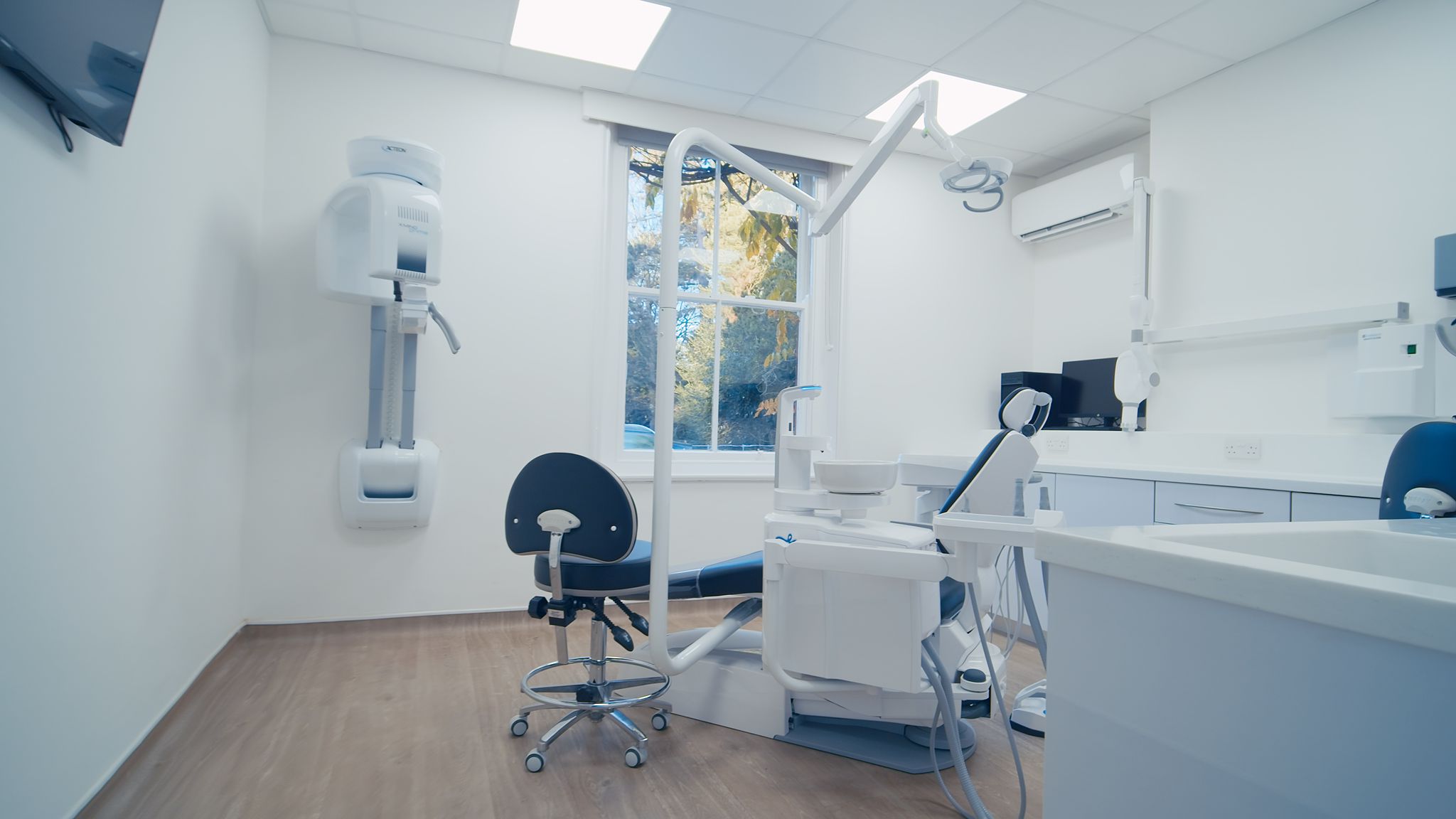
Acteon X-Mind Prime®, a genius extra-oral device that seamlessly combines beauty with practicality.

See what’s new with Planmeca at Dentistry Show 2024

Highest power without compromises!










